1. Introduction
On January 26, 2026, the “Immediate Market Entry Medical Technology System” came into effect, significantly shortening the time to clinical adoption for innovative medical devices and streamlining related procedures.
To implement the new system, the Ministry of Health and Welfare (“MoHW”) and the Ministry of Food and Drug Safety (the “MFDS”) amended the Rules on the Evaluation of New Health Technologies and the Regulations on Approval, Notification, and Review of Medical Devices.
Under the new system, new medical technologies that utilize medical devices having undergone internationally recognized clinical evaluation may be used in clinical practice without undergoing a separate New Health Technology Assessment. The reform is expected to accelerate market entry and enhance patient access to innovative medical devices.
2. Background and Implementation
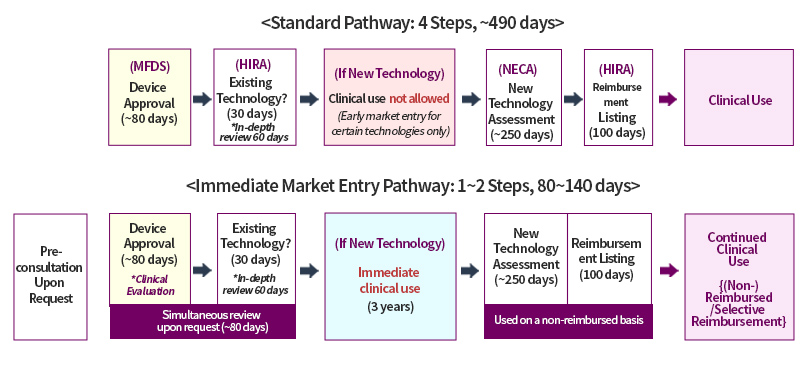
Under the standard market entry framework, medical devices are required to complete the following steps before market use: (i) approval by the MFDS (Step 1); (ii) confirmation by the Health Insurance Review and Assessment Service (“HIRA”) as to whether the technology of the medical device qualifies as an existing technology (Step 2); (iii) if confirmed not to be an existing technology, New Health Technology Assessment (“nHTA”) by the National Evidence-based Healthcare Collaborating Agency (“NECA”) (Step 3); and (iv) reimbursement listing by HIRA (Step 4). This process could take up to 490 days, with the nHTA under Step 3 alone requiring up to 250 days to assess the safety and efficacy of new medical technologies.
These lengthy timelines often delayed the market entry of innovative devices, raising concerns about reduced industry competitiveness. Although the government introduced measures to shorten review periods under a “pre-entry, post-evaluation” framework, practical limitations remained.
In response, the government has now introduced a pathway allowing immediate market entry, without a separate nHTA, for medical devices that use technologies other than existing technologies, provided they have already undergone enhanced clinical evaluation at an internationally recognized level. With this reform, eligible devices may enter the market immediately, potentially reducing the overall market entry period to as little as 80 days (the “Immediate Market Entry Pathway”).
[Ministry of Health and Welfare and Ministry of Food and Drug Safety, Press Release dated January 25, 2026]
3. Key Features of the Immediate Market Entry Pathway
The MoHW amended the Rules on the Evaluation of New Health Technologies to define eligible technologies and establish application and designation procedures, thereby allowing qualifying technologies to enter the market without a separate nHTA.
The MFDS amended the Regulations on Approval, Notification, and Review of Medical Devices to provide the basis for designating eligible device categories and to specify enhanced clinical evaluation data requirements. In consultation with the MoHW, the MFDS has designated 199 eligible categories, which includes the following:
- Digital medical devices (113 items): including stand-alone software and AI-based digital medical devices under the Digital Medical Products Act;
- In vitro diagnostic reagents (83 items): in vitro diagnostic reagents designated under the Medical Device Product Classification Notice for In Vitro Diagnostic Medical Devices; and
- Other devices: including robotic surgical systems, robot-assisted orthopedic exercise devices, and powered exoskeletons.
To use the pathway, applicants must submit internationally recognized enhanced clinical evaluation data to the MFDS at the MFDS device approval stage (Step 1) and obtain HIRA confirmation that the relevant technology is not an existing technology (Step 2). If the technology is confirmed not to be an existing technology, an application for immediate market entry (i.e., deferral of the nHTA) is deemed to have been filed. The Minister of Health and Welfare must then publicly announce the intended use, target patient population, procedure, and deferral period.
Once the nHTA is deferred through this process, the technology may be used immediately in clinical practice on a non-reimbursed basis for up to three (3) years. In light of potential patient cost burdens, the MoHW may, where deemed necessary, initiate an ex officio nHTA during the deferral period and determine reimbursement eligibility at an earlier stage.
4. Implications
The implementation of the Immediate Market Entry Pathway marks a significant shift toward a more market-oriented regulatory framework for medical devices in Korea. In particular, for sectors involving advanced technologies, such as AI-based digital medical devices, in vitro diagnostic devices, and medical robotics, the reform substantially lowers regulatory barriers that have historically delayed market entry. As a result, it is expected to have a direct impact on product commercialization, investment decisions, clinical evidence generation, and overall regulatory strategy.
Notwithstanding immediate market entry, the nHTA and reimbursement listing procedures will continue in parallel, and reimbursement determinations may, in some cases, be made earlier than anticipated. Accordingly, companies seeking to utilize the pathway should pursue not only an expedited market entry strategy but also a comprehensive plan for clinical evidence development, data management, and post-market evaluation.
Shin & Kim’s Healthcare and Life Science Practice Group comprises professionals with prior experience at key regulatory authorities, including the Ministry of Health and Welfare, the Health Insurance Review and Assessment Service, and the Ministry of Food and Drug Safety, working in close collaboration with attorneys specialized in medical device, digital health, and reimbursement regulations. We provide comprehensive legal advisory services across the entire regulatory lifecycle, including medical device approval strategy, response to New Health Technology Assessments, market entry structuring, and reimbursement and non-reimbursement strategy review.
Drawing on our regulatory insight and industry experience, we proactively assess risks arising from regulatory developments and deliver tailored legal solutions to support the swift commercialization and stable market integration of innovative medical technologies.
[Korean version] 시장 즉시진입 의료기술 제도 시행: 혁신적 의료기기 시장 진입 기간 단축






